Periodic Table of Elements Worksheets
About These 15 Worksheets
The periodic table might look like just a giant chart of boxes, but it’s really the ultimate cheat sheet for understanding matter. These worksheets break it down into fun, manageable lessons that help students see the table as a tool, not a wall of confusion. From learning about atomic numbers to exploring element families, kids get a guided tour of how this table organizes the building blocks of the universe. By practicing step by step, they learn to read, use, and even predict information from the table with confidence.
This collection covers everything from the basics to more advanced concepts. Some worksheets focus on simple recognition-like spotting metals versus nonmetals-while others dig into trends, isotopes, and electron configurations. Visual learners will love the color-coded versions, while analytical thinkers will enjoy exploring patterns and rules. With activities that range from classification to problem-solving, there’s something here for every learning style.
Most importantly, these worksheets show how the periodic table connects to real-world science. Understanding element properties and trends makes chemistry less mysterious and more practical. Whether students are preparing for a test or just starting to explore atoms and elements, this collection builds the foundation they’ll use throughout higher-level science classes. It turns the periodic table from intimidating to empowering.
Have a Look Inside Each Worksheet
A Blueprint of Matter’s Building Blocks
Students get introduced to the periodic table as a “blueprint” of all matter. The worksheet helps them see how elements are arranged and why it matters. It breaks the big, intimidating chart into manageable parts. Kids begin to see the table as an organized map of chemistry.
The Structure and Organization
This worksheet focuses on rows, columns, and element families. Learners discover how patterns make the periodic table logical instead of random. The activity feels like uncovering a code hidden in the chart. It strengthens understanding of how elements are grouped.
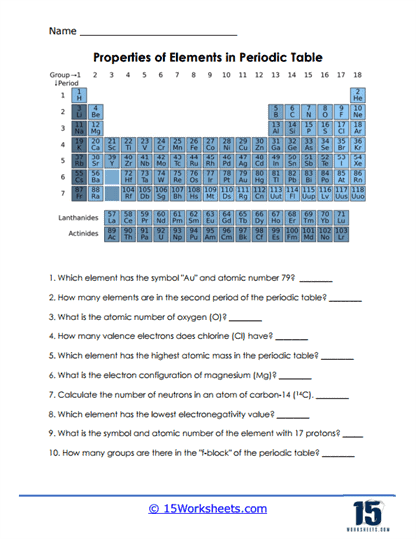
Properties of Elements
Students practice identifying whether elements are metals, nonmetals, or metalloids. The activity highlights differences in states of matter and other traits. It’s a great way to connect science facts to real-world properties. Kids learn that the table isn’t just numbers-it’s full of useful clues.
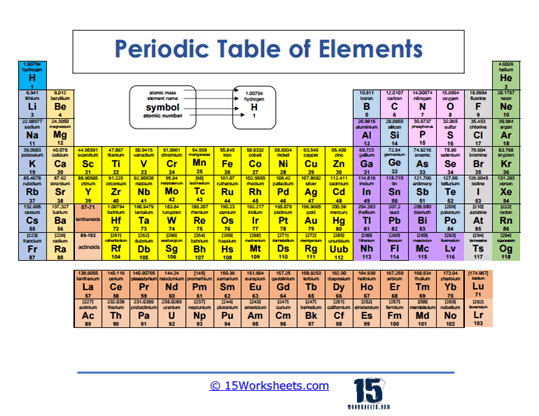
Black and White Periodic Table
Learners work with a clean, simple version of the table. The worksheet encourages them to focus on symbols, numbers, and placement. It strips away distractions so students can master the basics. This makes the foundation for more advanced activities later.
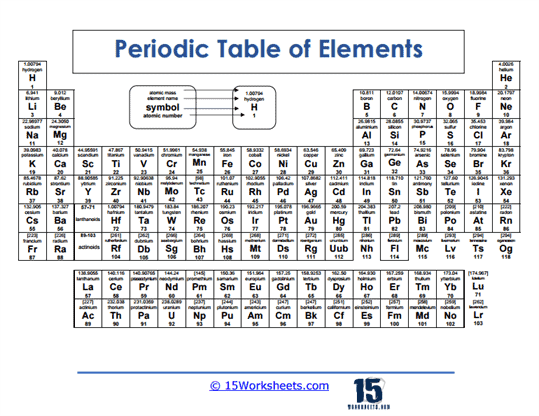
Colored Periodic Table of Elements
This worksheet uses color to show patterns like groups and element types. Visual learners benefit from seeing categories stand out. The activity makes the table more approachable and memorable. Students begin to recognize patterns at a glance.
Atomic Numbers
Students practice finding and using atomic numbers. The activity shows how these numbers define each element. It’s like giving every element its own ID card. Learners understand how atomic structure connects to the table.
Electron Configuration
This worksheet dives into how electrons are arranged in shells. Students see why elements behave the way they do in reactions. The activity feels like piecing together a chemical puzzle. It builds the link between atomic structure and real chemistry.
Using the Table
Students apply the periodic table to answer real questions about elements. It’s all about turning reference skills into problem-solving skills. The worksheet helps them use the table as a working tool. Kids gain confidence navigating the chart independently.
Organization of the Table
This worksheet explores why the table is set up the way it is. Learners look at periods, groups, and trends in detail. It’s like a history lesson mixed with science logic. Students learn that the design of the table tells a story.
Elements and Isotopes
Students explore the concept of isotopes within the periodic table. The worksheet shows how not all atoms of an element are identical. It introduces variety and complexity in atomic structure. Learners get a deeper understanding of how science classifies matter.
Periodicity
This activity highlights repeating trends across the table. Students practice spotting patterns in reactivity, size, and other properties. It feels like finding rhythms in chemistry. Kids learn that predictability is what makes the table so powerful.
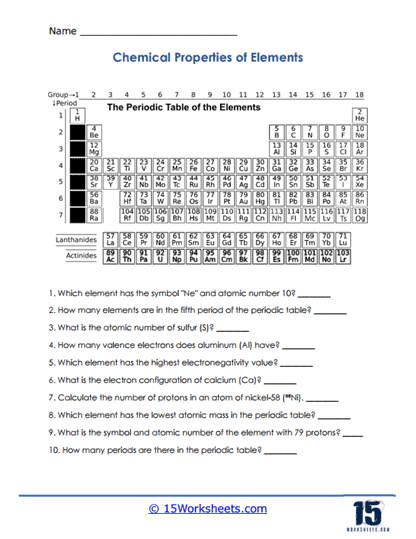
Chemical Properties of Elements
Learners focus on how different elements react and interact. The worksheet ties symbols and numbers to real-world chemical behavior. It makes the periodic table feel alive with possibilities. Students connect abstract data to concrete chemistry.
Rules in Classifying Elements
This worksheet covers the systems scientists use to organize the table. Students learn why certain elements belong in specific groups. The activity simplifies what can feel like memorization into logical rules. It strengthens both organization and comprehension skills.
Grouping Elements
Students practice identifying families such as noble gases, halogens, or alkali metals. The activity is like sorting elements into their “teams.” It makes classification hands-on and interactive. Kids build stronger recall of key element groups.
Periodic Trends
This worksheet explores trends like atomic radius, ionization energy, and electronegativity. Students learn how properties shift across rows and columns. It feels like mapping out invisible forces in chemistry. Learners begin to see the table as a predictive tool.
More Periodic Trends
Students dig deeper into additional patterns across the table. The activity gives extended practice with advanced trends. It pushes learners to think critically about relationships between elements. This builds readiness for higher-level chemistry.
Strange Trends
This worksheet looks at exceptions to the rules. Students see that science has quirks and surprises too. The activity makes learning more interesting by breaking the patterns. It reminds kids that even the periodic table has mysteries left to solve.
What is the Periodic Table of Elements?
The Periodic Table of Elements is a scientific chart that organizes all known chemical elements in a structured manner based on their properties. It was first developed by Russian chemist Dmitri Mendeleev in 1869, who arranged elements according to their atomic mass and observed that certain properties recurred periodically. This early version of the table has since evolved, and modern versions are organized by increasing atomic number, which corresponds to the number of protons in an element’s nucleus. The periodic table is a fundamental tool in chemistry, physics, and many other scientific fields, providing an easy reference to important information about elements, including their atomic structure, chemical behavior, and relationships with other elements.
The periodic table is arranged in rows, called periods, and columns, known as groups or families. Each period corresponds to a different energy level of electrons surrounding an atom’s nucleus. As you move from left to right across a period, the atomic number of the elements increases, meaning that each subsequent element has one more proton and one more electron than the previous element. While moving across a period, the chemical properties of the elements change progressively because of changes in electron configuration. On the other hand, elements within the same group (column) have similar chemical and physical properties because they have the same number of electrons in their outermost energy level, or valence shell. These outer electrons largely determine how an element reacts with others.
For example, Group 1 of the periodic table contains elements known as the alkali metals, like lithium (Li), sodium (Na), and potassium (K). These elements all have a single electron in their outer shell, making them highly reactive and prone to losing that electron in chemical reactions to form positive ions. Group 18, on the other hand, contains the noble gases, such as helium (He), neon (Ne), and argon (Ar). These elements have full outer electron shells, making them very stable and chemically inert under most conditions. This organization of elements into groups with shared characteristics makes it easier to predict the behavior of unknown elements or to understand the chemical behavior of compounds.
The periodic table is a powerful tool because it condenses a vast amount of information into a simple visual format. By looking at the table, one can quickly gather key facts about an element, such as its atomic number, atomic mass, and even its general chemical reactivity. In addition, the table reveals trends in properties, such as electronegativity, ionization energy, and atomic radius. For instance, elements become more electronegative as you move from left to right across a period, and they tend to become larger as you move down a group. These trends help scientists and students predict how elements will interact in chemical reactions, as well as explain the structure and properties of complex compounds.