Properties of Elements
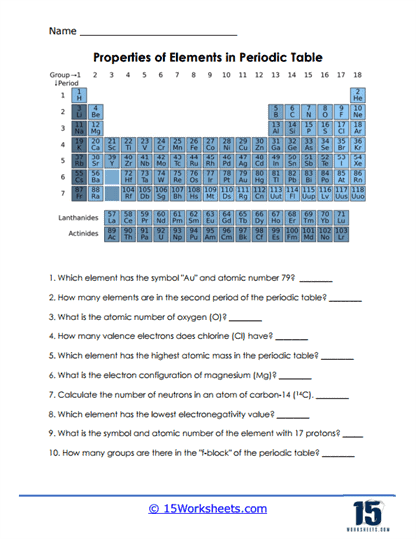
Worksheet Description
This worksheet offers students a detailed representation of the Periodic Table of Elements, accompanied by a series of questions that probe their understanding of the table’s various attributes. It delves into different facets of the Periodic Table, such as symbols and atomic numbers of specific elements, the number of elements in particular periods, electron configurations, and the atomic masses of elements. Additionally, there are questions about valence electrons, the concept of electronegativity, and the distribution of elements in various blocks of the table. The objective is to test a student’s depth of knowledge regarding the structure and characteristics of the elements on the Periodic Table.
To tackle this worksheet effectively, students should begin by familiarizing themselves with the layout and organization of the Periodic Table provided. They should understand how elements are classified into periods and groups and be able to identify elements based on their atomic numbers and symbols. For questions regarding atomic masses, valence electrons, and electronegativity, students will need to tap into their foundational chemistry knowledge and potentially use external references. Moreover, deducing the electron configuration or calculating the number of neutrons in a specific atom would require applying principles learned in chemistry lessons.
The worksheet is meticulously designed to enhance a student’s grasp on the diverse properties and attributes of elements present in the Periodic Table. It aims to foster both recall of basic facts and the application of deeper conceptual knowledge. The variety in question types ensures that students not only remember elemental specifics but also comprehend the broader patterns and classifications inherent to the table. Overall, this worksheet aspires to solidify a student’s understanding of the Periodic Table, making it a valuable tool for any budding chemist.
